
GA301 Product Description
1. Product Description:
This product is a high purity concentrated saponin adjuvant, can also be used as a general immune enhancer, the properties of light yellow or amber clarified transparent liquid, the main ingredient is a high purity immunoreactive substances, pH value of 4.0 ~ 6.5. safe and non-toxic metabolizable, able to effectively induce immune response of the organism, the T-cell-dependent and nondependent antigens have a very strong immune effect, the effect of mucosal immune response is remarkable.
2. Mechanism of action:
GA301 can enhance the differentiation and proliferation of immune cells in the immune organs, by increasing the number of natural immune cells, promoting the differentiation and maturation of natural immune cells, reducing the expression of immunosuppressive molecules and promoting the proliferation of immune effector cells, thus exerting its immune-enhancing effect. At the same time, GA301 and antigen enter antigen-presenting cells through cytosolization, and after GA301-mediated destruction of endosomal membranes, the cleaved protein antigen can be further processed into smaller peptide fragments in cytoplasmic lysate through proteasome mechanism. The degraded peptides translocate to the endoplasmic reticulum via transporter protein molecules, where they are facilitated to bind to newly synthesized MHC-I molecules and thus migrate to the cell surface via Golgi vesicles. Finally, peptide epitopes associated with MHC-I molecules exposed on the surface of DCs (dendritic cells) are presented to na?ve CD8+T cells via T cell receptors (cross-presentation). Provides a co-stimulatory signal to secrete cytokines and elicit an immune response.
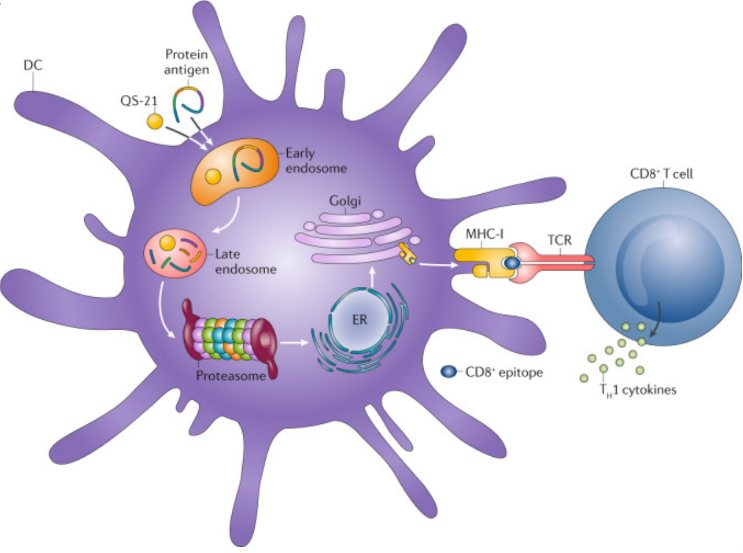
Ref: Carlo, Pifferi,Roberto, Fuentes,Alberto, Fernández-Tejada,Natural and synthetic carbohydrate-based vaccine adjuvants and their mechanisms of action.[J] .Nat Rev Chem, 2021, 5: 0.
3. Scope of application:
Applicable to all kinds of veterinary vaccines, including inactivated vaccines, genetically engineered subunit vaccines, etc. For example, it is recommended to be applied to circular vaccines and vaccines that need to be conveniently extracted or injected without needles.
Take several approved products as examples to illustrate the role of GA301 adjuvant:
3.1 Foot and mouth disease vaccine: GA301 adjuvant can induce efficient generation of specific antibodies against prevalent bacterial serotypes;
3.2 Porcine Reproductive and Respiratory Syndrome (PRS): Pigs can quickly resume growth after vaccination with GA301 adjuvant-containing vaccine, while viremia and clinical risk can be significantly reduced;
3.3 Porcine roundworm vaccine: water-soluble adjuvant for less irritation, improved piglet survival and reduced reproductive disorders in sows.
3.4 Schistosomiasis vaccine: GA301 adjuvant targeting the immunodominant peptide (plasmid-expressed) of the main Schistosomiasis mutant substantially reduces parasite loads in sheep;
3.5 Vaccine against toxoplasmosis: the rod-shaped protein + GA301 adjuvant stimulates humoral, local and systemic immune responses; this in turn partially protects the brain against the formation of crystals;
3.6 Dairy herpesvirus vaccine: a vaccine consisting of dairy herpes inactivated antigen + GA301 adjuvant induced high titers of antibodies (IgG of different subclasses), as well as an elevated DTH response was observed in mice; the level of cellular immunity against Th1, mediated by INF and IL-2, was also enhanced;
In summary, pet or veterinary vaccines recommended for use with GA301 adjuvant are:
Viruses: equine influenza, feline leukemia virus, Schmallenberg virus, bovine respiratory syncytial virus, bovine rotavirus, bovine coronavirus, parainfluenza 3 virus;
Bacteria: Borrelia mansoni, Coxiella burnetii;
Parasites: Cestodes, T. solium, E. granulosus;
4.Product Information:
Product Name: GA301
Product Code: GC07001
Grade: GLP grade, GMP grade
Packing specification: 50ml/vial, 100ml/vial (customization available)
