
3D-MPLA Product Description
GC Biotech’s 3D-MPLA is a detoxified derivative of the lipopolysaccharide (LPS) of Salmonella minnesota, strain R595, which retains the immunostimulatory activities of the parent LPS. It consists of a mixture of congeners, all containing a backbone of β1′→6-linked disaccharide of 2-deoxy-2-aminoglucose phosphorylated at the 4′-position, but differing in the fatty acid substitutions at the 2, 2′ and 3′ positions. The immunostimulatory activities of 3-O-desacyl-4′-monophosphoryl lipid A combined with the vaccine include up-regulation/down-regulation of co-stimulatory molecules on antigen-presenting cells and secretion of pro-inflammatory cytokines, resulting in an enhanced immune response of the Th1-type against the antigens. 3-O-desacyl-4′-monophosphoryl lipid A is a lyophilised powder or a sterile liquid.
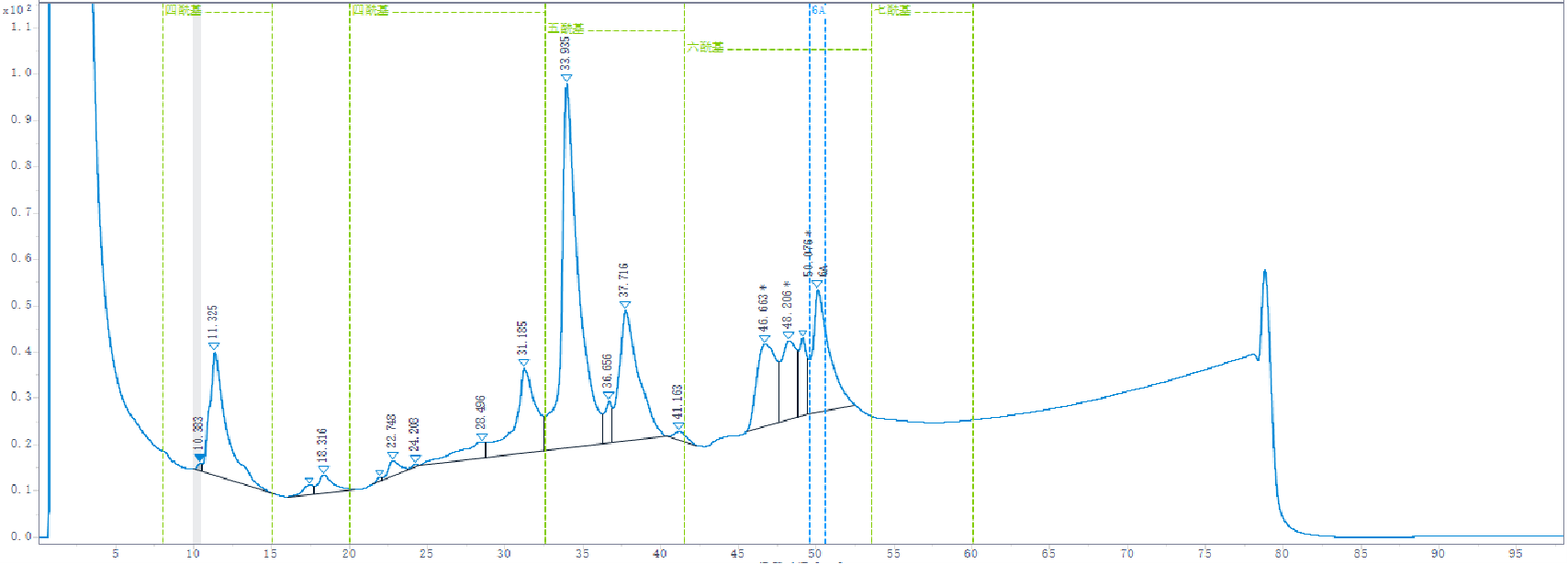
Our production method have been shown to yield consistently a 3-O-desacyl-4′-monophosphoryl lipid A comparable in structure and function with a preparation of 3-O-desacyl-4′-monophosphoryl lipid A used as adjuvant in the particular vaccine of proven clinical efficacy and safety in man.
The relative amount of each congener group in the triethylamine salt of our 3D-MPLA is:
– tetraacyl: 15 per cent to 35 per cent;
– pentaacyl: 35 per cent to 60 per cent;
– hexaacyl: 20 per cent to 40 per cent;
– heptaacyl: less than 0.5 per cent.
Now we supply Research / GMP grade 3D-MPLA to our clients.
Product Information:
Product Name: 3D-MPLA
Product Code: GC07003
Grade: GLP grade, GMP grade
Package specification: 5mg/vial, 100mg/vial (customization available)
